Abstract
There are a large number of drugs on the market that claim to be highly effective in combating stomach pain caused by reflux syndrome and increased hydrochloric acid. In practice, the efficacy of such drugs may vary. The purpose of this research work is to quantify the effectiveness of each of the four medications, Mylanta®, Gaviscon®, Tums®, and Omeprazole®, in the laboratory. The measure used was the number of drops of acid required to be added to a solution of each drug to change the color of bromocresol purple to yellow, i.e., to lower the pH. The results showed that Mylanta was the most effective drug, demonstrating maximum resistance to the action of hydrochloric acid, while Omeprazole showed results almost indistinguishable from those of distilled water. The paper’s findings highlight the importance of conducting thorough investigations and comparisons of drugs that claim high efficacy but cannot be confirmed in experiments.
Introduction
Hydrochloric acid is the primary component of gastric juice, typically serving a cleavage function; however, its imbalance can lead to various pathologies. Thus, one of the most common causes of abdominal pain is a gastric biochemical imbalance mediated by excessive hydrochloric acid secretion (Maret-Ouda et al. 2020). The causes of this condition are multiple and often lead to reflux and thus complicate the course of the disease at both symptomatic and physiologic levels.
Drugs that relieve pain and eliminate the causes of the ailment aim to neutralize hydrochloric acid in two strategies. On the one hand, such drugs contain antacids, which quench the acid by converting it into salts, lowering the pH of the stomach (Bailey 2017). On the other hand, drugs can stimulate the formation of a foamy layer that covers most of the stomach, serving as a barrier to prevent reflux (Bailey 2017; Lødrup et al. 2014).
There is a large number of drugs with varying efficacies in neutralizing hydrochloric acid, and the present laboratory work aims to conduct a quantitative study of this. To determine the efficacy of each solution, the present blind experiment utilizes the indicator bromocresol purple, which changes color to yellow in the presence of low pH. The addition of hydrochloric acid to the drugs enables the study of their efficacy in acid neutralization through visual examination. The present experiment hypothesizes that the substances in the spectrum used have different efficacies. Among the four drugs, there is at least one that is characterized by its maximum effect.
Materials and Methods
For background calibration, distilled water was used first, and 5 mL of it was transferred into a test tube using a pipette. Four drops of bromocresol purple were added to this tube. After thorough mixing, one drop of hydrochloric acid was introduced, and the resulting color changes were noted in the table as the baseline (control) level. Four drugs (A, B, C, D) were obtained from the teacher in different consistencies.
As a preparatory step, the tablet forms of the drugs were crushed using a mortar and pestle and dissolved with 100 mL of distilled water in a 250-mL beaker. The liquid forms of the drugs were carefully transferred into 250-mL beakers in single-dose sizes. One by one, 5 mL of each of the four drugs was transferred to a test tube with four drops of bromocresol purple added.
After vigorous stirring, one drop of hydrochloric acid was added to each. The results of the visual observation were recorded in a table, including the number of drops of acid required before the solution ceased to be purple and took on a yellowish hue. Additionally, the coloration of each solution was examined 10 minutes after all drops were added.
Results
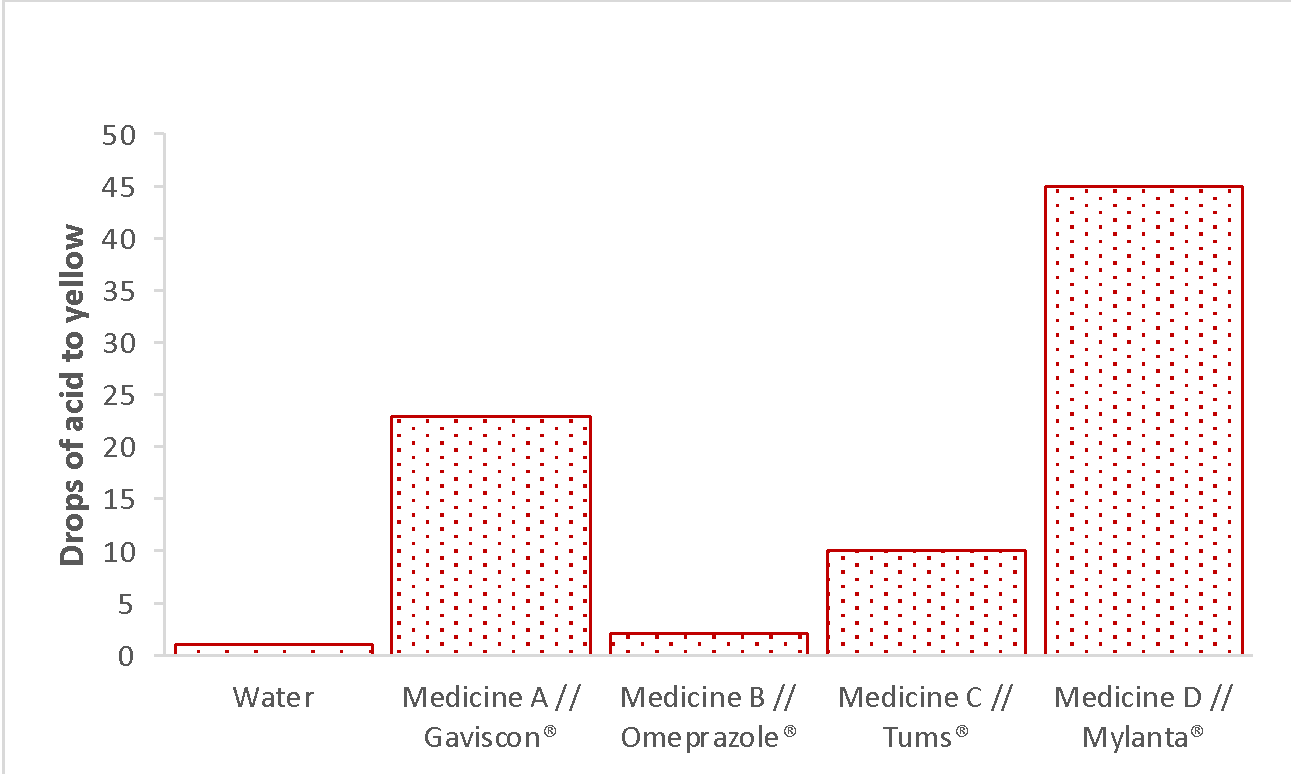
The results of the experiment were presented by the total number of drops of each solution and water required to acquire a yellow color. In addition, Table 1 lists the color of the solutions after 10 minutes of observation and the efficiency rating, which describes the efficiency of hydrochloric acid uptake by the solutions. As can be seen from the results, the drugs can be arranged in the following order: Mylanta® > Gaviscon® > Tums® > Omeprazole, with respect to their ability to neutralize hydrochloric acid, or, to put it differently, the number of drops of acid required to neutralize these drugs. The visual observation results showed a similar trend: only Omeprazole®, as the least effective drug, did not revert to purple coloration over time, while the top 3 drugs lost their yellow coloration, indicating successful acid neutralization.
Table 1. Results of the primary data for the experiment
Discussion
The present experiment aims to investigate the efficacy of four drugs in neutralizing hydrochloric acid, a component of gastric juice that can cause discomfort in patients with gastric imbalances. It is evident from the results that the greater the number of drops added, the greater the resistance of the substance to the acid. Thus, among all, Mylanta is the most effective neutralizer, followed by Gaviscon, Tums, and finally, the least effective Omeprazole, the quantification of which differs little from the outcomes for distilled water. The results thus support the initial hypothesis about the differing efficacy of commercial drugs.
The results obtained fit well with previously published evidence. It is widely known that proton pump inhibitors (e.g., Omeprazole) have therapeutic effects, whereas antacids (E.g., Gaviscon, Tums, Mylanta) are predominantly symptomatic for the ailment (Bailey 2017; Lødrup et al. 2014). This implies that the short-term efficacy of antacids is enhanced because they quickly eliminate the adverse effects of reflux, but do not promote long-term treatment, which is entirely consistent with the results that antacids inhibit acid to a greater extent. The use of a long-term analysis time (10 minutes) is insufficient in the context of the patient. Therefore, as part of a future study, it would be acceptable to observe the efficacy of these drugs over a longer time frame.
References
Bailey L. 2017. Advance directives and do not resuscitate orders. Family Doctor.
Lødrup A, Christina R, Peter B. 2014. Use of antacids, alginates, and proton pump inhibitors: a survey of the general Danish population using an internet panel.Scand. J. Gastroenterol. 49(9): 1044-1050.
Maret-Ouda J, Markar SR, Lagergren, J. 2020. Gastroesophageal reflux disease: a review. JAMA324(24): 2536-2547.