Introduction
Nitroaromatic compounds are biodegradable compounds in which, the NO2 group is attached directly to the cyclic aromatic structures; that can easily explode once exposed to explosion inducing conditions. In this respect, QSAR analysis for nitroaromatic compounds is the process through which; the compounds undergo different mutagenic reactions resulting into reduced compounds, referred to as hydrolamines. Throughout the process of reaction, the nitroaromatic compounds correlate with their mutagenic activity which yields molecular energy. More specifically, during such reactions; the determination of the energies of the respective LUMO and HOMO molecular orbital is the key position, necessary to provide a basis for comparison between the two reactions (Mati 2000).
Analysis of QSAR 1 using logs TA 100 and log P
As it usually happens in any chemical reaction involving nitroaromatic compounds, QSAR are established using regression analysis of the variables involved. On this basis therefore, QSAR 1 results can be used to deduce that log TA 100 is likely to correlate well with just log P; as any change in log TA 100 results into a corresponding change in the log P. However, this correlation between the log TA 100 and log P is negative; since any increase in the log TA 100 leads to a decline in the log P that proceeds to very low values including negatives (Devillers, 2009).
From the QSAR’s 1-3, the multiple R undergoes an activity which is satisfactorily with various individual properties. Based on the observations made, correlation coefficient 0.6 is the best as it gives the lowest negative value among the other correlations indicating that; the reaction will have yielded the best results possible. Further, the multiple R has strong correlation with log P, EEUMO and ELUMO reactions which results into the ultimate correlation of the three variables (Spain 1995).
The relationship between EEUMO and ELUMO reactions
As it has been observed from QSAR’s 1 and 4, the quadratic function of P is not better than a linear function because as the outcome depicts; the representation of the results using the quadratic function of log P and linear function would give similar interpretation. More precisely, the use of the quadratic function of log P and that of linear equation would have no difference in giving the inference over the reaction involved (Devillers, 2009).
Further, for QSAR’s 4 and 5 a correlation with EEUMO is better than E HOMO because from the observations made in the experiment; the reactants involved had the lowest negative correlation coefficient which definitely indicated a better correlation in EEUMO than as it is in E HOMO. Meanwhile, the F stat is affected in various ways in which; the significance of the correlation was largely determined by the size of F. For instance, the F stat in the reaction was affected by the amount of reactants involved and the conditions under which the reaction took place; like the pressure and the temperature levels (Matsumoto 2000).
Ring structures with more than two fused rings
As it was observed, some particles were poorly predicted because they were seen to lie far from the line of prediction. For example, the particles of 3-nitro-fluoxenone and 2-acetoxy-7-nitrofluorene were observed to have a significant difference in the predicted and the observed values of correlation. From the experiment it was also found that, various ring structures had more than two fused rings of various kinds in which; they shared edges as shown in the diagrams below (Devillers, 2009).
By assigning a value of I=1 for each of these particles and I=0 for the other systems, majority of the molecules with I=1 predicted were more active than found experimentally. On the other hand, majority of those molecules with I=0 predicted were less active than found experimentally. Perhaps, the incorporation of the indicator value helped to improve the correlation to a greater extent whereby; the variables involved exhibited a better and a more eligible degree of reactivity. For example, when NH-OH was exposed to sulfotransterase became oxidized to NAc-OSO3H which gave a higher value of correlation coefficient than without the indicator (Devillers 2009).
More so, the findings above could also be explained in terms of the type of Salmonella typhimurium strain used in the experiment. As it was observed, the strain contained nitroreductose that was activated into groups at various rates in different conditions. From the experiment, the strain was observed to be induced to intercalate with DNA by various substances (Kontogiorgis & Pontiki 2005).
Improvement of reactions involving EEUMO and ELUMO
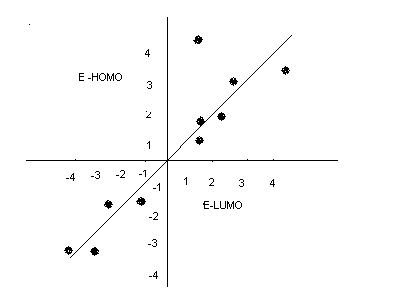
From QSAR 7, the inclusion of E HOMO improved the quality of the fit as it can be seen that; the correlation became better with the inclusion of the E HOMO. Generally, the E HOMO inclusion in the reaction helped improve the quality of the reaction which further enhanced the achievement of better results that were easy to interpret. The graph below shows the comparison between the ELUMO and E HOMO (Brinkley 1999).
Conclusion
As it can be seen in the graph, EEUMO and EHOMO are directly related in the sense that; an increase in the E HOMO leads to a consequent increase in the ELUMO and vise versa. From the results obtained it can be inferred that, any other related chemical reaction to the one observed in the experiment would have similar results. More specifically, the results obtained could be used to predict the results of another related activity by studying closely the effect of each input on various variables and the output obtained.
Reference list
Brinkley, D., 1999. Thermal and Photocatalytic Oxidation of 2-Propanol on Aromatics. Washington: University of Washington Press.
Devillers, J., 2009, Endocrine Disruption Modeling (QSAR in Environmental and Health Sciences). New York: CRC Press.
Kontogiorgis, A. & Pontiki, E., 2005. A Review on Quantitative Structure-Activity Relationships (QSARs) of Natural and Synthetic Antioxidants Compounds. New York: Bentham Science Publishers.
Mati, K., 2000. Molecular Descriptors in QSAR/QSPR including CD-ROM. New York: Wiley-Interscience Publishers.
Matsumoto, A., 2000. Surface Materials Having Nitrogen Oxide Removing Function. Hirata Kogyosho: Japan Patent Publishers.
Spain, J., 1995. Biodegradation of Nitroaromatic Compounds: Review of Microbiology, New York: Plenum Press.