Abstract
This laboratory report examines the relationship between environmental parameters and ethanol yield as a product of yeast metabolic fermentation. Yeast is microscopic fungi that produce alcohol from glucose under the influence of enzymes. This is an essential biochemical reaction that is often used in science, industry and cooking because the ethanol produced has valuable properties for communities. In addition, the product ethanol is a pure substance of natural origin, for which reason it is often referred to as bioethanol. In the present virtual experiment conducted in a Labster environment, variables such as temperature, pH, stirrer, and airflow were manipulated to investigate their ultimate effect on alcohol yield.
The fermentation process took place in a nitrogen atmosphere. It was shown that the yeast was very sensitive to changes in pH, as its decrease or increase resulted in a lack of reaction initiation. The highest yield was possible only under initial initiation conditions – later, this was compared with scientific data. A good correspondence was found between the detected parameters and the reference figures, which means that the current work agrees well with the scientific information.
Introduction
Under natural conditions, yeasts of the genus S. cerevisiae can produce ethanol as a product of metabolic activity. Complex carbohydrates, which are carbon sources for yeast, undergo alcoholic fermentation, resulting in the formation of monatomic alcohol. Because of its natural origin, ethanol is often called bioethanol, emphasizing the mechanism of molecule formation. Obviously, the ethanol yield is highly dependent on the environmental conditions under which the yeast is active. This laboratory report examines the dependence of bioethanol production on temperature, pH, stirring, and gas composition.
Methods
Labster, an academic online service, was used to perform the virtual experiment. In this environment, independent variables, including temperature (°C), pH, stirrer (%), and airflow (L/min) were manipulated using a dedicated interface. The initial settings were set as shown in Table 1.
Table 1. Parameters of the different experiments that were used in the work.
Results
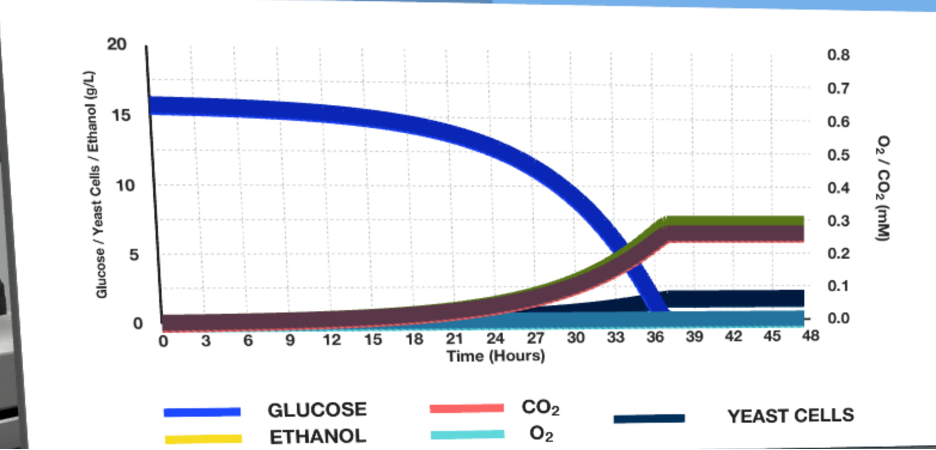
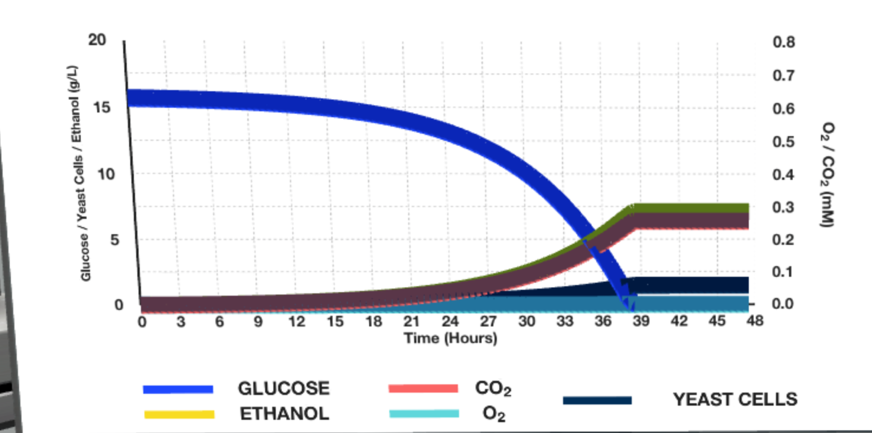
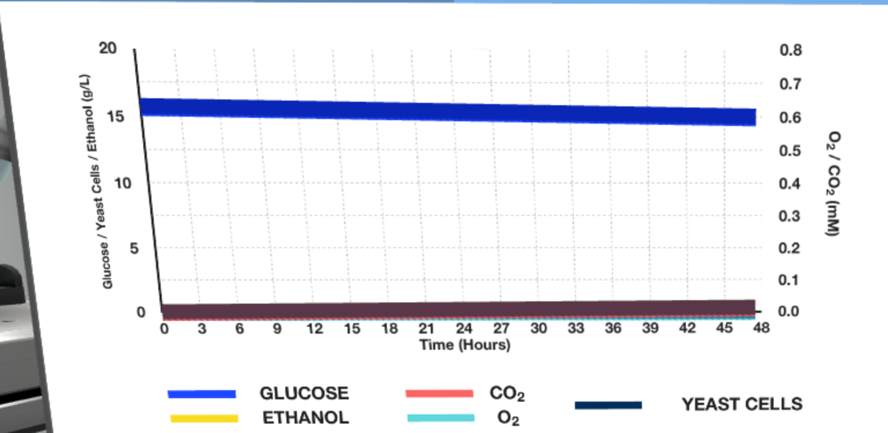
Running the fermenter at the given plant parameters resulted in the curve plots shown in Figures 2-3 below.
Discussion
A key feature of yeast is the ability of micro fungi to produce ethanol produced by the metabolic fermentation of glucose. More specifically, under the influence of glycolysis, glucose inside yeast cells is converted into pyruvate, which produces two molecules of acetaldehyde when carbon dioxide is stripped off. In turn, acetaldehyde is converted to ethanol by enzymatic action; the entire process is accompanied by the release of ATP energy (Guglielmo, 2018).
Figure 1 clearly shows that the glucose and ethanol concentration curves are almost reversible: over time, the amount of glucose decreases sharply, and the amount of alcohol increases during the first day, after which it gradually decreases. The maximum ethanol production, in this case, is reached only at the 36th hour and does not exceed 7-8 g/L. Figure 2 shows that an increase in temperature by five °C corresponded only to a shift in the time of maximum yield, increasing it by 5-6 hours.
Figure 3 shows that changing the pH to two while maintaining other parameters does not trigger the enzymatic reaction. Consequently, it is reasonable to assume that the optimal temperature for ethanol production is 30°C, and the pH should be 4-6. This fits well with publicly available data indicating the need for 26°C at a pH of 4-4.5 (Brooke, 2021; Hamel, 2019). Thus, suitable conditions were found to be relatively reliable.
References
Brooke, C. (2021). Which pH is optimum for yeast fermentation from 4-8? The Whole Portion. Web.
Guglielmo, T. (2018). Fermentation. Microreviews in Cell and Molecular Biology, 4(2), 1-3.
Hamel, P. J. (2019). Where to put dough to rise. King Arthur Baking. Web.
Appendix A