Definition of the Underlying Concepts
A weak acid is one in which ions dissociate partially in water. A strong base is a compound that can remove a proton from a significantly weak acid. Weak acids include acetic acid, formic acid, and benzoic acid. Strong bases comprise various compounds such as sodium hydroxide, potassium hydroxide, and calcium hydroxide (Libretexts, 2022). When a weak acid is titrated with a strong base, a basic solution is produced at the equivalent point. In this case, the equivalence point will be a pH 7, although it does not show exactly standing at the seven mark.
The basic solution is provided so that the anion of the acid becomes a common ion that lowers the ionization of the acid. Weak acid and strong base mixture result in the following net-ionic equation HA+ OH⁻ → A⁻+ H₂O (Abeka, 2013). It is important to indicate if the two are equimolar; the PH can be arrived at by considering the equilibrium reaction of A- with water.
Understanding Why Titrating a Weak Acid with a Strong Base Results in a Basic Equivalence Point
In the titration curve, the solution acts as a buffer. Hence, the base will overcome the capacity of the buffer. The titrate mixture is a salt solution at the equivalence mark since the acid cannot donate hydrogen ions (H+) to water, whereas the base accepts H+ from water. All weak acid is neutralized and changed to their conjugate base. At this point, the moles in H+ equals the number of moles in OH- (Libretexts, 2022). At the equivalence point, the chemical formula is indicated H30=OH- and the splitting of water molecules in the reaction between water and ions of salt occurs during hydrolysis.
Additionally, it is important to mention that the weak acids react with the equivalent portion of the strong base at the equilibrium point. Hence, the solution contains a salt of a weak acid and a strong base, whereby the salt is a basic solution. To elaborate further, the weak acid chemically reacts reversibly with water where hydronium ions are formed, and the acid is at the conjugate status.
Graphical Explanation of the Titration of a Weak Acid with a Strong Base
In a titration graph, the equilibrium curve goes slightly to the left (Libretexts, 2022). After the equilibrium, the titration process ejects hydronium ions towards the right, whereby a weaker base is realized, as shown in Figure 1 (Abeka, 2013). The equilibrium point involves a reaction of all acids where a solution with a relatively large amount of weak base is left.
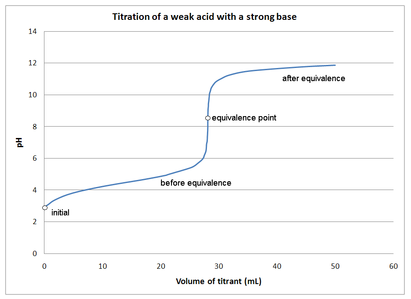
This reaction can be shown using titration for acetic acid with sodium hydroxide. This can be shown in a chemical equation as follows; HC2H3O2 + OH– → H20 + C2H3O2– (Abeka, 2013). During that titration, the 0H- reacts with H+ in the acetic acid to form an acetate ion, as shown in the equation. That means the conjugate base has reacted with water to form a slightly basic solution. Acid and base titrations usually depend on neutralization between the two compounds when mixed in a solution (Libretexts, 2022).
The endpoint for the equivalence point may not be the same since the equilibrium point is affected by the reaction’s stoichiometry. At the equivalence point, normally, the pH is greater than seven due to acid conversion to a conjugate base (Abeka, 2013). It is important to determine the initial pH so that values derived can be accurate since titration may have a determinant factor that may influence the result.
References
Abeka. (2013). Chemistry: Precision and Design (3rd ed.). A Beka Book.
Libretexts. (2022). Acid-base titrations. Chemistry LibreTexts. Web.
Supriya, N. (2022). What is acid-base titration theory? Definition, types, video & procedure. Biology Reader. Web.