Introduction
Hepatocellular carcinoma (HCC) is a common form of cancer affecting approximately one million people annually (Savci, 1999). The incidence of this liver cancer has increased over the years even though this cancer is generally regarded to be a malignancy of the aged because of its’ high prevalence (El-Serag, 2001). Studies to date have identified cirrhosis of the liver and infections from the hepatitis C and B virus as the key risk factors for the development of HCC (Velazquez et al., 2003). HCC has a poor prognosis, thus a favorable outcome is largely dependent on early diagnosis because there are more curative treatment options to select from. However, early diagnosis is often not achieved because the disease often progresses without distinct signs or symptoms. The most frequently reported non-specific symptom for HCC is abdominal pain and weight loss (Kuba et al., 2006). Similar to other forms of cancers, prognosis for HCC is heavily influenced by tumor stage and the extent of liver function because the latter can affect the patient’s tolerance to the different treatment regimes (Llovet et al., 1999). Curative management strategies following diagnosis of HCC include liver transplant, tumor ablation therapies and surgery, the latter is the method of choice provided the selection criteria is met (Kuba et al., 2006). Curative tumor ablation therapies include methods such as radiation therapy and cryosurgery (Geschwind et al., 2003), ethanol percutaneous injection, thermotherapy and chemotherapy (Aguayo & Patt, 2001). Although there are several curative strategies to choose, in actuality less than fifteen percent of patients are cured with one or more of the above methods (Kuba et al., 2006). Although prognosis is poor, overall, considerable progress has been made in recent years due to advances management techniques and advances in field of medical imaging. Undoubtedly, a crucial factor for long-term HCC survival is early and correct diagnosis and staging since there are fewer treatment options when the disease has progressed and is at an advanced stage. Regardless of whichever imaging modalities is used, the ultimate aim is to identify and characterize both the benign and malignant tumors so patients are appropriately managed. Selection of the optimal imaging technique is not always an easy task since they all differ in sensitivities and specificity (Fernandez & Redvanly, 1998). Of the different imaging modalities available on the market today, CT and MRI are among the favorite for imaging of the liver given that ultrasounds are limited to the detection of larger HCC tumors (i.e. greater than 2cm) (Peterson et al., 2005). The ability to detect small tumors or lesions is important when a curable outcome is desired. This issue is complicated because of two major factors; the first is due to low contrast between normal tissue and the tumor. The second factor is because smaller size lesions are less likely to be detected regardless of whichever imaging modality is employed (Savci, 1999). Two reliable imaging tools reported by Choi and coworkers (2001) and confirmed by Arguedas et al. (2003) include the triple-phase helical CT and triple phase contrast enhanced MRI. This paper will discuss the diagnostic value of imaging modalities on the identification of HCC and will focus in particular on x-ray computed tomography (CT) and magnetic resonance imaging (MRI). In addition, the future directions of HCC diagnosis will also be explored.
Principles of MRI
MRI uses a powerful magnetic field and hydrogen protons inside the body to create images. These protons align with the direction of the field when a patient goes inside the powerful magnetic field of the MRI. The protons release energy at a radio frequency which can be detected by the scanner when the field is switched off. These emissions are picked up and made into digital images using computers.
It important to note that imaging for diagnostic purposes is usually done so as to help the medical practitioner to differentiate between a normal tissue and a diseased tissue. The difference between a normal tissue and a tissue with a disease can be noticed easily by contrast on the images. Contrast simply means the difference in intensities of images and thus results are achieved when image density is high. Generally, contrast enhanced MIR helps in improving visualization of various tissues under study. The contrast of MIR depends on time of longitudinal relaxation which is usually denoted as T1. It also depends on transverse relaxation time of the tissue which is usually denoted as T2. Soft tissues that are not infected with diseases have different values of T1 but their values of spin density have very small variations. Thus T1-weighted imaging is one of the effective ways of obtaining good images. Changes in the values of T2 usually give good characteristics of most diseases and therefore T2-weighted is considered to be a very sensitive way of detecting diseases.
Diagnostic Accuracy of MRI
Although CT is highly recommended for liver imaging, a number of studies have suggested MRI is in fact, slightly better at diagnosing and characterization HCC lesions (Burrel et al., 2003; Libbrecht et al., 2002). MRI as an imaging modality is advantageous for a number of reasons. Firstly, it uses magnetic forces rather than X-rays and so there is none of the dangers associated with radiation exposure to. Secondly, this modality also has a higher specificity than CT (94% vs. 84%), even when no contrast agent was used (Dujardin et al., 2008). Thirdly, it can distinguish between HCC and other lesions. There is general agreement that the appearance of HCCs can vary and this is particular true on T1 weighted MRI images (Low, 2007). As a result, several studies have examined the potential benefits of using contrast agents such as gadolinium for liver imaging. When gadolinium is used, HCCs are heterogeneously enhanced in the arterial phase whereas benign lesions show typical enhancement, cysts exhibits no enhancement and haemangiomas displays discontinuous peripheral enhancement (Semelka and Helmberger, 2001). Images from the arterial phase must be captured between 16 to 20 seconds following gadolinium injection (Low, 2007). Fourthly, this method is still extremely useful because adverse reactions caused by contrast agent in patients are rare and reduced volumes of this agent can be used effectively (Kanematsu et al., 2006), for this reason some people consider this to be a better imaging option than CT. However, there appears to be some limitation in the use of MRI for HCC diagnosis. For instance, there is some disagreement in the literature regarding the sensitivity of gadolinium enhanced MRI for identifying small lesions. For example, a study conducted by Krinsky and coworkers in 2001 concluded gadolinium enhanced MRI was unable to accurately detect lesions of less than 2cm. However, another study conducted in 2006 by Hecht et al. reported gadolinium enhanced MRI was accurate in diagnosing HCC and suggested this method is accurate enough to be used on its own. Another technique that has shown promising results is the use of a specific liver contrast agent known as super paramagnetic iron oxide (SPIO). Following its administration, this chemical is taken up by Kupffer cells in the liver and the result is that malignant tumors appear hyperactive intense compared to normal liver tissues. Thus, SPIO enhanced MRI is recommended for imaging suspicious liver cancers since it is more sensitive than contrast enhanced CT at detecting malignant lesions (80-97% vs. 72-75%) (Reimer et al., 2000). Furthermore, several studies have reported superior diagnosis and characterization of HCCs when both contrast agents (gadolinium and SPIO) are used compared to the single use of either one agent (Lutz et al., 2005). Although MRI has proven to be beneficial in diagnosing and characterizing HCCs, its use is contradicted in a number of instances. For example, in patients possessing internal metal objects such as pacemakers and clips because MRI machines generate very strong magnetic fields. In addition, more patient cooperation and longer examination bookings are needed because abdominal MRI requires more time compared to CT scans. These must also be kept in mind when determining the diagnostic value of MRI. An example shown in figure1 is hepatocellular carcinoma detected by MRI.
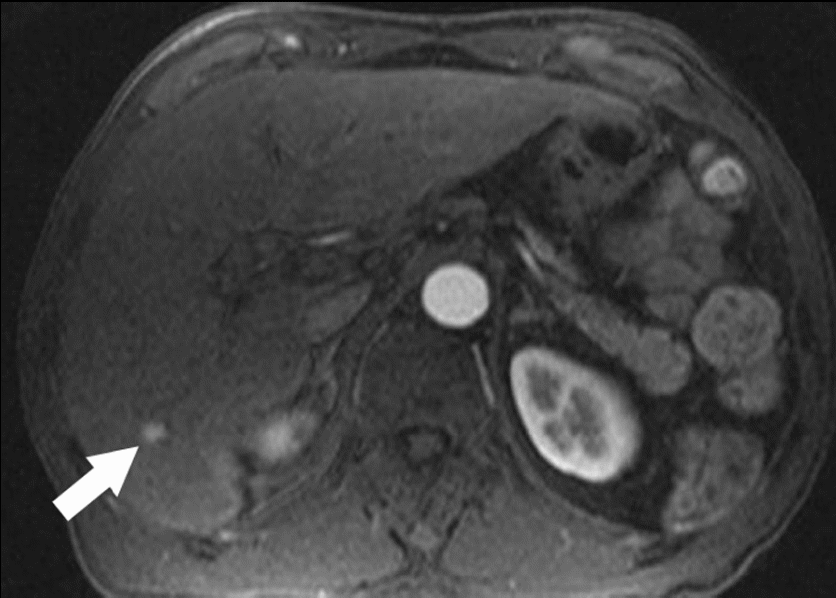
Principles of CT
In brief, CT images rely on X-rays being passed through the patient’s body and for this information to be collected by detectors on the other opposite side, the interconnected X-ray source and detector must be rotated around the patient during the scanning period. Cross sectional image known as a tomogram are then produced on digital computers using this data. Different tissues absorb X-rays differently because some tissues are more permeable than others. This is the reason why different tissues usually appear different during CT when forming X-ray films. Moreover, Lipiodol CT is very sensitive for detecting HCC.
There are two types of scanners that are commonly used in CT. The first one is sing-slice scanner which usually has a slice width of approximately 1mm. The width of slice is normally determined by the geometry of a scanner and the dimensions of its focal spot (Sato et al. 2000). Development has been made in improving single-slice scanner to multi-slice scanner so as to improve the efficiency of utilizing X-ray beams especially those in the Z-direction. The improvement has also been done to increase the speed of rotation of the X-ray tube and the whole system used for detection of the rays. In multi-slice scanner, the scanner width is reduced from 1mm to 0.5mm and the collimation of X-ray is extended in Z-direction. Detector arrays which have about 8 to 34 rows are used to make multi-slice scanners more effective.
Studies have also shown that there is direct correlation between densities of micro-vessels of renal cell carcinoma which is usually abbreviated as RCC and dynamic contrast TC. Contrast enhancement was heterogeneously distributed among the tumors that were studied. It is recommended that to effectively evaluate the effects caused by antiangiongenetic therapy, a whole tumor should be scanned rather than scanning a tumor in a single level. This is because the effect of antiangiogenetic varies depending on the location of the tumor. Multiphase scanning at an interval of every 9.5 seconds can be done in a single level or a single-phase scanning of 35 seconds delayed scanning can be done so as to compare the images of tumor obtained using the two methods. However, the radiation dose is higher when multiphase scanning is used. It is because of higher radiation dose that that multiphase scanning should be handled with care. In addition, CT is very useful for medical practitioners in evaluation of the degree of angiogenesis.
Diagnostic accuracy of CT
CT fluoroscopy is helpful to localize small lesions. While conventional CT often fails to indicate tumor location, Lipiodol spots on CT improve the likelihood of diagnosis (Sato et al. 2000). In addition, Lipiodol CT was useful to assess the tumor ablatory effect (Sato et al., 2000). At present, to detect hypervasucular HCCs 10 mm in diameter, Lipiodol CT is the most sensitive tool (Sato et al., 2000). An example shown in Figure 2 hepatocellular carcinoma was identified by Lipiodol CT. The efficacy of interventional radiographic CT fluoroscopy for CT-guided PEI was recently reported (Sato et al. 2000). However, because of the high radiation exposure, it should be used only when tumors are not delineated on Lipiodol CT. Therefore, Lipiodol injection is the essential step (Sato et al., 2000). However, further improvement in CT and MR technology might eliminate the use of Lipiodol in the future (Sato et al., 2000). Helical CT scanning has also improved the ability to detect HCC which is usually a hyper vascular tumor, by allowing acquisition of both hepatic arterial dominant and portal venous dominant sets of images during separate breath holds. There is no optimal timing that has been determined for a set of images dominated by arteries and the timing is also influenced by factors such as the size of the patient and status of cardiovascular (Murakami et al., 2001). Many investigators initiated arterial phase imaging by making use single detector row CT with a scanning delay of 20–30 seconds, but this series was usually completed within 40–50 seconds (Murakami et al., 2001). As with MRI the inherent risks associated with CT must also be borne in mind when analyzing the diagnostic value of CT in HCC diagnosis. For example, the use of radiation has been known to cause cancers (Murakami et al., 2001). In addition, the contrast agents used in this modality can cause severe, and sometimes life threatening, allergic reactions (Murakami et al., 2001).
In the latest studies that were done as described above, CT was performed in 28 patients who were suffering from metastatic liver disease and the results were summarized as follows: All the metastases showed low values of tissue densities when plain scan was used. A second examination using an intravenous medium was done on twenty patients among the initial twenty eight patients and 8 cases showed an increase in contrast difference. 4 cases showed a decrease in contrast difference while there was no noticeable change in the other 8 cases (Sato et al. 2000). The result of “No difference” in the density of a tissue can be detected when conducting CT with a contrast medium or without it.
Future directions of imaging in HCC
In the future there are some indications to suggest that MRI become a more valuable diagnostic tool in HCC imaging for two important reasons. Firstly, improvements in magnetic field strength will allow for even better quality imaging. Secondly, there is much current research being carried out into tissue-specific agents which is likely to provide favourable outcomes in HCC imaging in the near future (Choi, 2004). Indeed, Choi (2004, 25) claims that such contrast agents will increase the specificity and sensitivity MRI in HCC imaging.
Conclusion
In conclusion, the significant value of imaging modalities relating to HCC and the role of CT and magnetic resonance imaging has been highlighted. This is an area, which is being addressed around the world due to the many millions of people affected this condition. Furthermore, the alternative to these techniques is CT or US guided biopsy, which whiles it, may arguably provide better specificity and sensitivity for HCC diagnosis, it is both invasive and carries the danger of tumor seeding. It can be said that both CT and MRI are key factors in diagnosing tumor to the liver, and assisting in the future management of radiation therapy for the patient. This disease is particularly complex with some research indicating that better diagnostic results are available thought CT scan, while other research suggests that MRI is more helpful in detecting such masses present in the liver due to the high contrast factor. Since this liver disease remains to be potentially life threatening in many cases, it is hoped that further research and developments into medical imagining will continue in order to aid in early detection and therefore improve prognosis.
References
Aguayo, A., and Y. Z. Patt. 2001. Nonsurgical treatment of hepatocellular carcinoma. Clinical Liver Disease 5: 175-189.
Arguedas MR., Chen VK., and Eloubeidi MA. 2003. Screening for hepatocellular carcinoma in patients with hepatitis C cirrhosis: a cost-utility analysis. American Journal Gastroenterology 98: 679-690. Web.
Bruix, J., M. Sherman, J. M. Llovet, M. Beaugrand, R. Lencioni, A. K. Burroughs, E. Christensen, L. Pagliaro, M. Colombo, and J. Rodés. 2001. Clinical Management of Hepatocellular Carcinoma. Conclusions of the Barcelona-2000 EASL Conference. Journal of Hepatology 35 (3): 421-430.
Burrel M., Llovet JM., Ayuso C. et al. 2003. MRI angiography is superior to helical Ct for detection of HCC prior to liver transplantation: an explants correlation. Hepatology 38: 1034-1042.
Choi D., Kim SH., Lim JH. et al. 2001. Detection of heptacellular caccrcnoma: combined T2-weighted and dynamic gadolinium-enhanced MRI versus combined CT during arterial portography and CT hepatic arteriography. Journal of Computer Assisted Tomography 25: 777-785.
Dujardin M., Vandenbroucke F., Boulet C., Op de Beeck B. and de Mey J. 2008. Indications for body MRI Part I. Upper abdomen and renal imaging. European Journal of Radiology 65: 214-221. Web.
El-Serag HB. 2001. Epidemiology of hepatocellular carcinoma. Clinical Liver Disease 5 (6): 87-107.
Fernandez MDP. and Redvanly RD. 1998. Primary hepatic malignant neoplasms. Radiological Clinic North America. 36: 333-348.
Geschwind JF., Ramsey DE., Choti MA., Thuluvah PJ. and Huncharek MS. 2003. Chemoembolization of hepatocellular carcinoma: results of a metaanalysis. American Journal of Clinical Oncology 26: 344-349.
Hecht EM., Holland AE., Israel GM. et al. 2006. Hepatocellular carcinoma in the cirrhotic liver: gadolinium-enhanced 3D T1-weighted MR imaging as a stand-alone sequence for diagnosis. Radiology 239: 438-447.
Kanematsu M., Kondo H., Goshima S., Kato H. et al. 2006. Imaging liver metastases: Review and update. European Journal of Radiology 58: 217-228. Web.
Krinsky GA., Lee VS., Theise N.D. et al. 2001. Hepatocellular carcinoma and dysplastic nodules in patients with cirrhosis: prospective diagnosis with MR imaging and explantation correlation. Radiology 219: 445-454. Web.
Kuba D., Valdes D., Uribe M. and Sanchez N. 2006. Hepatocellular carcinoma. An overview. Annals of Hepatology 5 (1): 16-24. Web.
Libbrecht L., Bielen D., Verslypoe C. et al. 2002. Focal lesions in cirrhotic explants livers: pathological evaluation and accuracy of pretransplantation imaging examinations. Liver Transplant 8: 749-761.
Llovet JM., BruA C. and Bruix J. 1999. Prognosis of hepatocellular carcinoma: the BCLC staging classification. Seminars in Liver Disease 19: 329-339.
Low R. 2007. Abdominal MRI advances in the detection of liver tumours and characterization. Lancet Oncology 8: 525-535.
Lutz AM., Willmann JK., Goepfert K. et al. 2005. Heptatocellular carcinoma in cirrhosis: enhancement patterns at dynamic gadolinium-and superparamagnetic iron oxide-enhanced T1-weighted MR imaging. Radiology 237: 520-528. Web.
Murakami, T., T. Kim, M. Takamura, M. Hori, S. Takahashi, M. Federle, K. Tsuda, K. Osuga, S. Kawata, and H. Nakamura. 2001. Hypervascular hepatocellular carcinoma: Detection with double arterial phase multi-detector row helical CT 1. RSNA.
Peterson J., Bancroft L. and Kransdorf M. 2005. Principles of tumor imaging. European Journal of Radiology 56: 319-330.
Reimer P., Jahnke N., Fiebich M. et al. 2000. Hepatic lesion detection and characterization: value of nonenhanced MR imaging, superparamagentic ion orside-enhanced MR imaging, and spiral CT-ROC analysis. Radiology 217: 152-158.
Sato, M., Y. Watanabe, K. Tokui, K. Kawachi, S. Sugata, and J. Ikezoe. 2000. CT-guided treatment of ultrasonically invisible hepatocellular carcinoma. The American journal of gastroenterology 95 (8): 2102-2106.
Savci G. 1999. The changing role of radiology in imaging liver tumours: an overview. European Journal of Radiology 32: 36-51.
Semelka RC. and Helmberger TKG. 2001. Contrast agents for MR imaging of the liver. Radiology 218: 27-28. Web.
Velazquez RF., Rodriguez M., Navascues CA. et al. 2003. Prospective analysis of risk factors for hepatocellular carcinoma in patients with liver cirrhosis. Hepatology 37: 520-527.