One of the emerging issues in health care is the prevention of unnecessary re-hospitalization of patients diagnosed with certain conditions such as heart failure (Verhaegh et al., 2014). In the recent past, a number of strategies have been implemented by hospitals to reduce healthcare costs and offer quality patient care. The proposed research study is aimed at understanding the effectiveness of transitional and non-transitional care models towards reducing readmission rates in elderly patients with heart failure. Before undertaking the research, several steps and action plans will be undertaken in order to achieve the targeted goals (Zamanzadeh, Valizadeh, Tabrizi, Behshid, & Lotfi, 2015). Such action plans will be considered in order to ensure a professional and ethical study is conducted. As indicated in phase 2, the research will target 40 heart failure patients receiving healthcare support from a medical institution.
Pre-Implementation Phase
Seeking Ethical Clearance
The first step to consider before implementing the project is getting the right ethical clearance. The relevant ethics committee (EC) will be informed about the targeted study. The committee will be updated about the procedures, steps, and expectations of the research project. This clearance will ensure the study is completed in accordance with the stipulated ethical guidelines (Zamanzadeh et al., 2015). It is also necessary to note that the committee will be updated frequently throughout the study period. The EC will also be provided with a detailed summary of the findings after the research is completed successfully.
Seeking Informed Consent from Participants
As mentioned earlier, the study will target forty elderly patients who have received medical care from a health institution. One of the fundamental requirements of every study is protecting the confidentiality and anonymity of the participants throughout the process. Every participant will be provided with a form detailing the goals of the study. The participants should be sure that the research will accord the required privacy to them (Franks, 2015). This means that the implementers and healthcare practitioners will not disclose the information of the patients. This kind of approval is required in order to encourage the participants to provide quality information that can support the research study.
Budget and Resources
The success of the targeted study will be determined by the availability of the right resources and finances. To begin with, competent individuals will be recruited to be part of the Quality Improvement (QI) team. The members of the QI team will be recruited from the healthcare institution. The role of this team will be to assess the rate of admissions and results of various medical interventions (Verhaegh et al., 2014). This analysis will be conducted for the two groups of patients. Writing materials and computers will be acquired to ensure the gathered data is recorded in a timely manner.
The projected budgetary allocation for the proposed study will be around $20,000. The money will be used to purchase the required resources such as computers, journals for different entries, and writing materials (Zamanzadeh et al., 2015). The money will also be used to cater for transportation costs. The targeted participants will also receive gifts such as chocolates and handkerchiefs in order to feel motivated. Members of the QI team will receive medical equipment such as thermometers and gloves. These resources will play a positive role towards ensuring that the study is successful.
Initiating the Implementation Process
Several players will be involved throughout the implementation phase of the project. Members of the research team such as healthcare workers, the QI team, and stakeholders will be part of the process. The leaders will review of objectives, hypotheses, and goals of the project (McHugh & Ma, 2013). During the stage, every emerging issue or concern will be addressed accordingly. The concept of teamwork will be considered in order to ensure every activity is within the adjusted budget and/or timeline (“The Centers for Disease Control and Prevention,” 2014).
Documentation of Every Process
Throughout the implementation phase, a number of processes and activities are undertaken by different players. That being the case, it will be necessary to document such processes in a timely manner (Verhaegh et al., 2014). This approach will ensure the participants are aware of the steps undertaken and eventually provide quality data. These questions will be used to underpin the project implementation documentation process: What is the scenario being studied and what is expected after completing the study?
Training of the Implementers
The success of the proposed study will be dictated by the capability and willingness of the implementers. The QI team will gather the relevant data based on the outcomes of the participants. This means that various sampling and documentation processes will be used during the study. Members of the QI team will therefore be trained in order to be aware of the targeted procedures. The leaders of the study will mentor the implementers and equip them with the required competencies. Monitoring will also be done throughout the research period. The other important thing to consider is that training will be an ongoing process during the implementation phase (Verhaegh et al., 2014). This approach will be undertaken in order to ensure the implementers and participants are guided throughout the process.
Pretesting
The next step that will be taken seriously during the implementation phase of the project is pretesting (Franks, 2015). The researcher does not plan to use special tools or equipment to conduct the study. This is the case because the ultimate goal of the research will be to gather and review participants’ data before, during, and after implementing the outlined interventions. During this step, the research team will examine the effectiveness and clarity of the research instructions. Any change that can improve the quality of the study will be considered during this stage (“The Centers for Disease Control and Prevention,” 2014). The QI team will also design appropriate forms that can be used to record the gathered information. The team will outline the procedures required to tabulate the gathered data.
Availing Resources
The study project cannot be completed successfully without equipping the stakeholders with the right resources. This step will be undertaken before executing the study. The QI team will receive the required writing materials, books, and computers. The stakeholders will also be consulted in order to ensure the required materials are purchased in a timely manner (Verhaegh et al., 2014). These procedures will play a key role towards ensuring that the research study is conducted successfully.
Data Collection and Management
The next critical step of the implementation phase is data management. In order to complete the study successfully, the targeted data will be collected by competent individuals. Documentation will also be done accurately by the QI team. The “use of appropriate information collection methods will make it easier for the scholars to trace data to their originality” (Zamanzadeh et al., 2015, p. 416). The implementers will be required to consider a number of characteristics in order to collect good data. Some of these characteristics will include accuracy, indelibility, and legibility (Franks, 2015). The next process will be to ensure the collected data is archived properly. This initiative will make it easier for the researchers to use the data in the future.
Time Frame of the Project
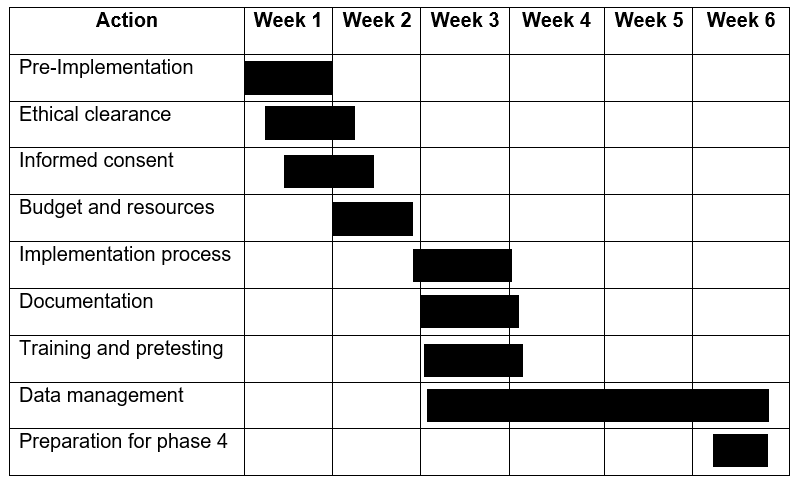
Concluding Remarks
The completion of the above steps will ensure the implementation phase is executed in a professional manner. The implementation process will be reviewed continuously in order to address the major issues that might affect the research project. Since the targeted elderly patients might not provide accurate information during the study, the QI team will be encouraged to monitor and support the needs of the participants (Zamanzadeh et al., 2015). Members of the family will also be involved during the process. This approach will make it easier for the patients to receive evidence-based medical support (McHugh & Ma, 2013). The conclusions from the research will be eventually used to come up with powerful healthcare practices that can reduce the rate of readmission in patients with heart failure. The insights gained from the completed study will guide healthcare practitioners to provide evidence-based medical support to more elderly patients with various terminal conditions.
References
Franks, S. (2015). Transitional care to reduce 30-day heart failure readmissions among the long-term care elderly population. Web.
McHugh, M., & Ma, C. (2013). Hospital nursing and 30-day readmissions among Medicare patients with heart failure, acute myocardial infarction, and pneumonia. Med Care, 51(1), 52-59.
The Centers for Disease Control and Prevention. (2014). Planning, implementing, and evaluating an intervention: An overview. Web.
Verhaegh, K., MacNeil-Vroomen, J., Eslami, S., Geerlings, S., de Rooij, S., & Buurman, B. (2014). Transitional care interventions prevent hospital readmissions for adults with chronic illnesses. Health Affairs, 33(9), 1531-1539.
Zamanzadeh, V., Valizadeh, L., Tabrizi, F., Behshid, M., & Lotfi, M. (2015). Challenges associated with the implementation of the nursing process: A systematic review. Iranian Journal of Nursing and Midwifery Research, 20(4), 411-419.